How To Extract Chlorophyll Using Coffee Filters
I take been having a discussion on another forum, on how it is possible for h2o to pickup chlorophyll in an extraction, when chlorophyll is basically a hydrocarbon, which is more often than not insoluble in h2o.
I did enough inquiry to know that I was in over my caput with chemistry that I took on the tardily fifties and early on sixties, so I asked Joe, our budding biochemist, to accept a run at it.
In quick summary, before Joe'south response, "define soluble?" The word soluble ways unlike things in biochemistry, than it does in organic chemistry, because of the behavior of the molecules of life.
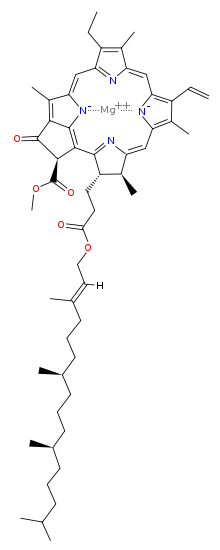
The chlorophyll molecule has a magnesium (Mg) at its rings centre, which makes it ionic and water-loving (hydrophilic) and a ring that is water fearing (hydrophobic) with carbonyl groups near a tail that go far polar (likewise hydrophilic).
It is held in place in the plant within a water-soluble cloth known as water-soluble chlorophyll-binding protein (WSCP). WSCP is soluble in water, and by and large insoluble in polar alkane alcohols and non polar alkanes.
Chlorophyll is readily soluble in alcohol, mostly insoluble in not polar alkanes like butane and hexane, and has some special relationships with polar water, considering of its polar and ionic groups.
Mostly is a cardinal word in all cases, because of chlorophylls charged polar finish and not polar hydrocarbon ring with the ionic Mg.
Before I finish summarizing, here is Joe'due south response to the question of how water can remove and transport basically insoluble chlorophyll molecules, as well as how saturating water with NaCl table salt aids in the process of washing unwanted chlorophyll out of extractions that have gone awry:
Chlorophyll Info by Joe
Chlorophyll is an intra-membrane chemical inside a thylakoid. A thylakoid is a membrane-bound compartment inside chloroplasts. 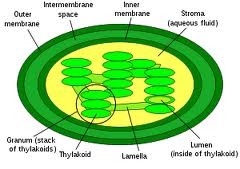 The thylakoid membranes of higher plants are composed primarily of phospholipids and galactolipids that are asymmetrically arranged along and beyond the membranes.
The thylakoid membranes of higher plants are composed primarily of phospholipids and galactolipids that are asymmetrically arranged along and beyond the membranes. 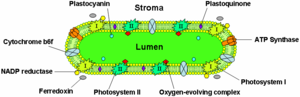
Chlorophyll is shown every bit photosystem I and II in this illustration.
Both phospholipids and galatolipids have hydrophilic (water-loving) heads and hydrophobic (water fearing) tails.
In biology this theme is used in virtually all life forms to compartmentalized for free energy storage, isolate invaders or encase their genome to protect information technology and many others reasons.
Solubility is a term that has more ane definition. In inorganic chemistry it refers to waters (or another compound) power to break covalent and ionic bonds of about compounds, dependent on time temperature and pH. The "solublized" atoms are so bonded to their polar opposite ion Hthree (+) or OH (-) and are in solution.
In biological science withal, it is used in the beginning tense merely, information technology is also used to describe the ability of an organic molecule or complex to form an association with water and exist in solution just not exist "solubilized" by information technology. Proteins and other organic molecules use charged ions such equally phosphate (PO4) and Sodium (Na) to grade micelles. Micelles are trivial balls of hydrophobic molecules surrounded by a charged ion.
Simply like a jail cell's membrane bilayer. Sometimes micelles are formed by complexes of proteins surrounding a modest molecule for transport through water.
Chlorophyll specifically, is merely able to course complexes with other molecules to stay in solution at biological pH (7.iv). Its natural surroundings is at a pH of around 4 non 7.iv. At this pH information technology has a cyberspace charge of -ii and so that information technology tin can class a chemo-gradient for electron transport during photosynthesis.
So since pH =-log [OH-/ H+] when at pH 4 the [H+] concentration is college than the [OH-] thus creating an environment that is more probable to acquaintance with the (-) charged area of the chlorophyll molecule. Hence the low solubility of unbound chlorophyll in water, the large hydrophobic areas compress together and nowadays their hydrophilic areas to exclude h2o from the eye, condign a mass that will sediment in h2o.
The point of this is to illustrate that while purified chlorophyll is not likely to stay in solution in pure water; nosotros don't extract pure chlorophyll and we don't use pure water.
We utilise alkali to proceed the charge on the phospholipid bilayer (Na+ with POfour -three) and no detergents. The alcohol (ROH+) wants nothing to do with Na+ while in its protonated (H+) land.
Non polar solvents for obvious reasons won't either and too won't course much of an emulsion with alcohols in their bent country considering; the alcohol is denser and forms a micelles like layer to protect itself from the charged Na+. If there is an excess of alcohol it will offset forming an emulsion layer at the upper interface.
The phospholipid bilayer of the chloroplasts and of the thylakoid existence intact or mostly and then, prevent the chlorophyll from being disassociated with the Na+ water and are able to be excluded from alcohol or non-polar solvents .
Alcohol is able to associate with chlorophyll and proteins in its native conformation just non when bent past Na+ because the electron pool concentrated at the (O-) repels the (-) region of the chlorophyll molecule and the PO4 -iii of the membrane bilayer.
If the membranes accept been broken upwards past a detergent or broken downwards by enzymes then the only fashion to exclude chlorophyll from a non-polar solvent or booze is with lots of Na+ and water. Because of the large not polar area of the chlorophyll molecule it tin can more easily grade a hydrophobic interface and shield the accuse in the heart.
The salt exposes the accuse (considering ionic is a stronger bond than Van Der Wals forces) and precipitates the chlorophyll into water.
And then in conclusion, chlorophyll is not wholly soluble in water, but in its biological circuitous is able to associate with it.
By manipulating charge/charge interactions chlorophyll tin exist forced into solution with h2o and away from polar organic and not-polar solvents. While it doesn't meet the inorganic chemistry definition of solubility, it will class micelles complexes with an ionic solution and it tin be precipitated from that solution nether the right weather.
From a biological perspective chlorophyll tin can also be solubilized by any solvent under the correct weather.
Sooooo, back to my summary, it appears that some solvents tin launder away the cement bounden the chlorophyll in the constitute cells and costless the chlorophyll to be done away as a micelles, but it also exposes the chlorophyll to the solvent, which in the example of alcohol, will readily dissolve information technology and concord it in solution.
Freezing the cloth prior to an extraction, may exist holding both the chlorophyll and the WSCP locked up in water ice, so that neither the water present or the solvent can reach the chlorophyll.
Merck Index lists chlorophyll as practically insoluble in non polar solvents, so the departure between practically and totally insoluble may offer a clue, besides as butanes practically insolubility in water.
While near sources list due north-Butane as insoluble in water, its actual solubility is 0.0325 vol/vol, at 1 temper pressure and 20C/68F. That is 32 ml/liter, which may exist plenty to business relationship for the light electric green hue that occurs, by both washing abroad the WSCP and belongings some of the chlorophyll in break.
When nosotros saturate the h2o with salt, before washing a polar extract suspended in a non polar solvent like hexane, information technology forces the chlorophyll into solution and washes it away.
Explanation by Joe, summarized by Graywolf
How To Extract Chlorophyll Using Coffee Filters,
Source: https://skunkpharmresearch.com/chlorophyll-pickup-in-extractions/
Posted by: childsrecare68.blogspot.com


0 Response to "How To Extract Chlorophyll Using Coffee Filters"
Post a Comment